Pack of 6 H2o Kayaks M5 Neoprene Well Nut Inc Stainless Steel Pozi Screw (Pack 6) Long Type D: Amazon.com: Industrial & Scientific

The structure diagrams of (H2O)6⁻ cluster isomers. The red and white... | Download Scientific Diagram

Use the balanced equation to answer each question. 2 C2H6(g) + 5 O2(g) delta 4 CO(g) + 6 H2O(g) How - Brainly.com
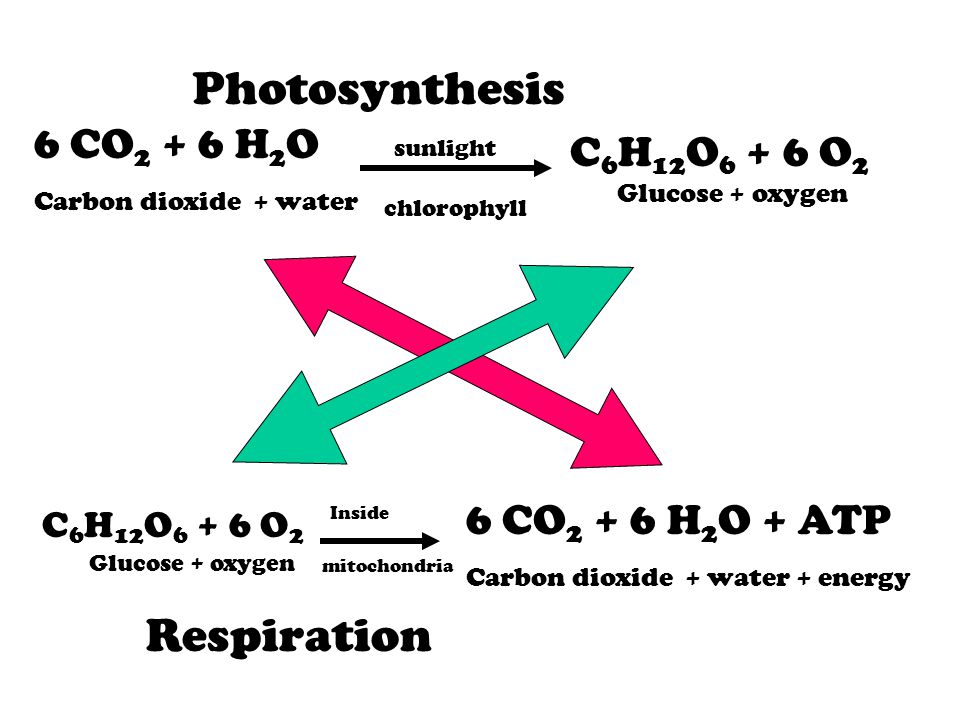
How to balance the Photosynthesis Equation (CO2 + H2O —> C6H12O6 + O2 + H2O? THE BALANCED EQUATION WILL BE OPTION A) 6 CO2 + 9 H2O —> C6H12O6 + 3 H2O + 6 CO2 OPTION B) 6 CO2 + 12 H2O —> C6H12O6 + 6 O2 + 6 H2O
![SOLVED: NiCl2·6 H2O + 3 C2H8N2 → [Ni(C2H8N2)3]Cl2 + 6 H2O Nick, Ethyl, and Amy reacted 3.60 g of NiCl2·6 H2O with 8.50 mL of 4.0 M C2H8N2and obtained 2.83 g of [ SOLVED: NiCl2·6 H2O + 3 C2H8N2 → [Ni(C2H8N2)3]Cl2 + 6 H2O Nick, Ethyl, and Amy reacted 3.60 g of NiCl2·6 H2O with 8.50 mL of 4.0 M C2H8N2and obtained 2.83 g of [](https://cdn.numerade.com/ask_previews/e7bf0f40-8942-4c62-a0e8-90258b6ac48b_large.jpg)
SOLVED: NiCl2·6 H2O + 3 C2H8N2 → [Ni(C2H8N2)3]Cl2 + 6 H2O Nick, Ethyl, and Amy reacted 3.60 g of NiCl2·6 H2O with 8.50 mL of 4.0 M C2H8N2and obtained 2.83 g of [
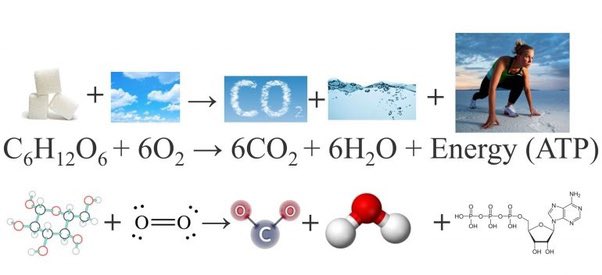
عبدالرحمن العتيبي on Twitter: "A simple example of a combustion reaction is the burning of methane: CH4(g) + 2 O2(g) → CO2(g) + 2 H2O(g)" / Twitter
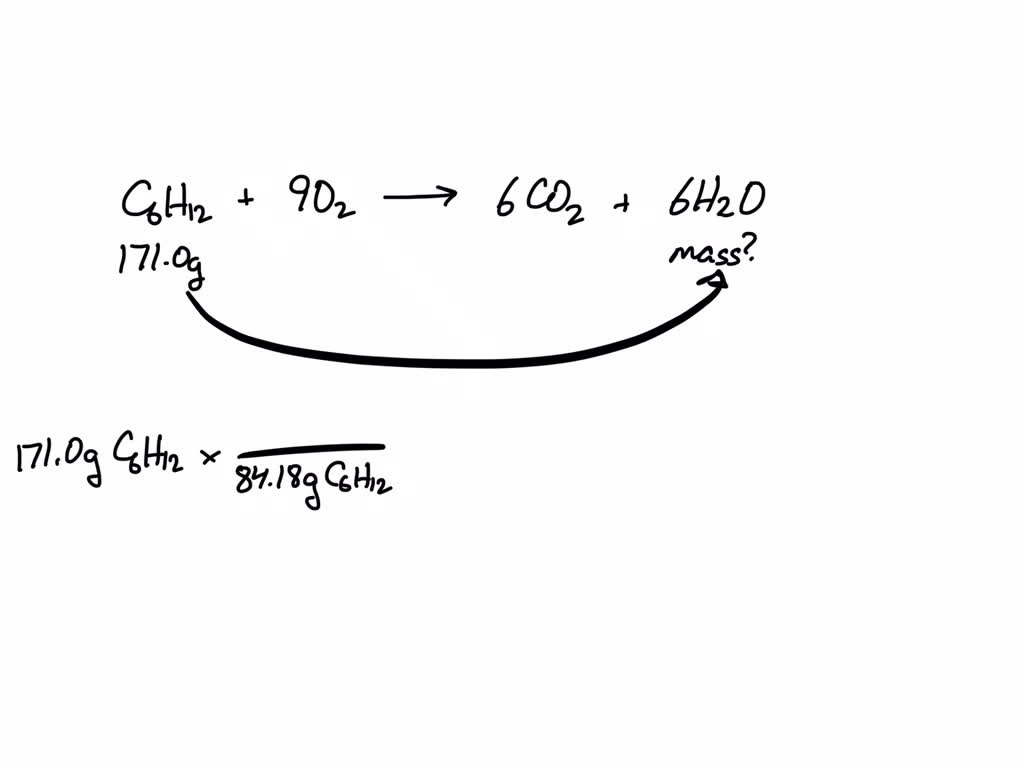
SOLVED: Use the following chemical equation C6H12 + 9 O2 —-> 6 CO2 + 6 H2O (OR C6H12 + 9 O2 right arrow 6 CO2 + 6 H2O) How many grams of
![Ground and Excited States of the [Fe(H2O)6]2+ and [Fe(H2O)6]3+ Clusters: Insight into the Electronic Structure of the [Fe(H2O)6]2+–[Fe(H2O)6]3+ Complex | Journal of Chemical Theory and Computation Ground and Excited States of the [Fe(H2O)6]2+ and [Fe(H2O)6]3+ Clusters: Insight into the Electronic Structure of the [Fe(H2O)6]2+–[Fe(H2O)6]3+ Complex | Journal of Chemical Theory and Computation](https://pubs.acs.org/cms/10.1021/ct501143c/asset/images/medium/ct-2014-01143c_0017.gif)
Ground and Excited States of the [Fe(H2O)6]2+ and [Fe(H2O)6]3+ Clusters: Insight into the Electronic Structure of the [Fe(H2O)6]2+–[Fe(H2O)6]3+ Complex | Journal of Chemical Theory and Computation
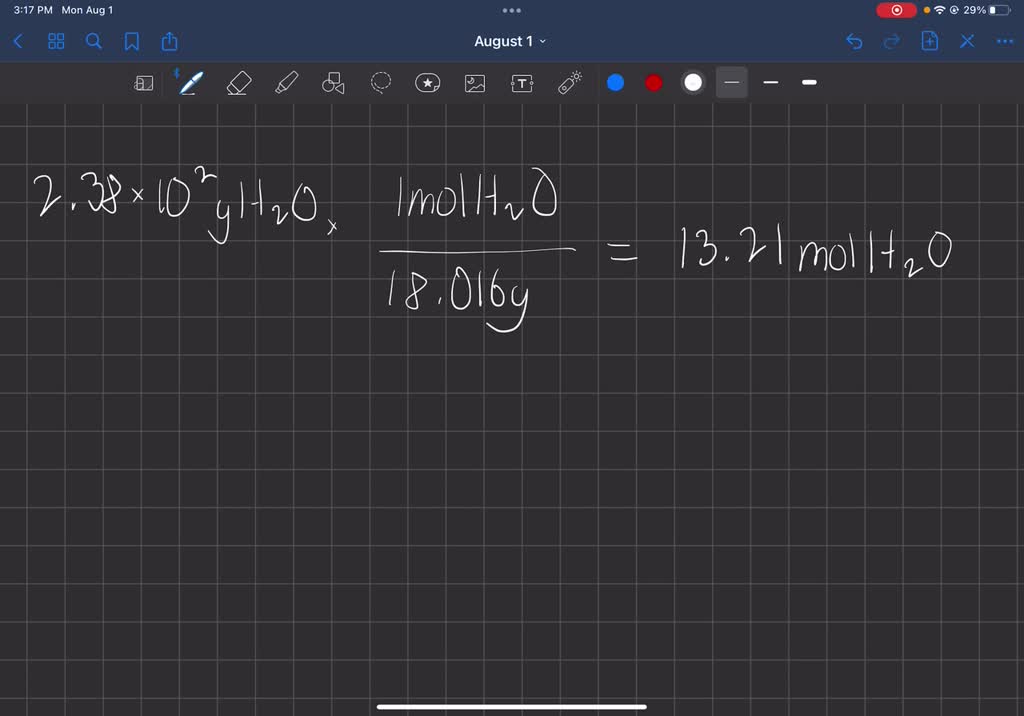
SOLVED: Consider this reaction: 6 CO2 + 6 H2O + light C6H12O6 + 6 O2If there were 2.38 x 102 g of H2O, 18.6 moles of CO2, and plenty of light, what would be the theoretical yield of the reaction?